- 翰林提供學術活動、國際課程、科研項目一站式留學背景提升服務!
- 400 888 0080
Edexcel IGCSE Chemistry 復習筆記 3.2.3 Catalysts
Edexcel IGCSE Chemistry 復習筆記 3.2.3 Catalysts
Catalysts & Rates
- Catalysts are substances which?speed up?the?rate?of a reaction without themselves being?altered?or?consumed?in the reaction
- The mass of a catalyst at the beginning and end of a reaction is the?same?and they do not form part of the equation
- An important industrial example is iron, which is used to catalyse the?Haber?Process?for the production of ammonia
- Iron?beads?are used to increase the?surface?area?available for catalysis
- Normally only?small?amounts?of catalysts are needed to have an effect on a reaction
- Different processes require different types of catalysts but they all work on the same principle of providing an?alternate route?for the reaction to occur
- They do this by lowering the activation energy required, hence providing a reaction pathway requiring less energy
- Catalysis is a very important? branch of chemistry in commercial terms as catalysts increase the rate of reaction (hence the production rate) and they reduce energy costs
- The transition metals are used widely as catalysts as they have variable oxidation states allowing them to readily?donate?and?accept?different numbers of electrons. This is key to their catalytic activity
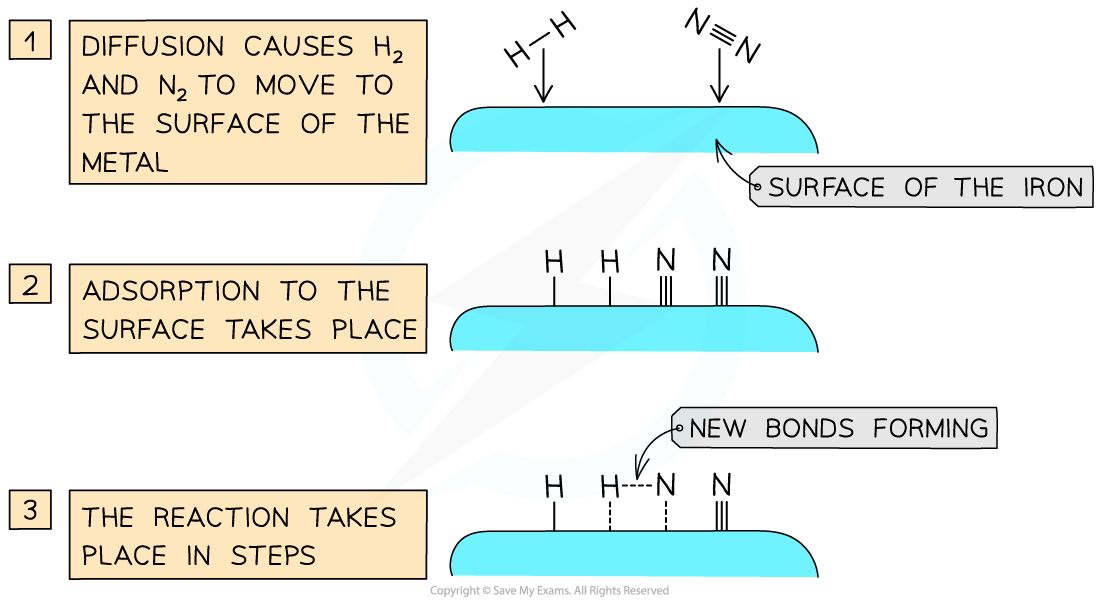
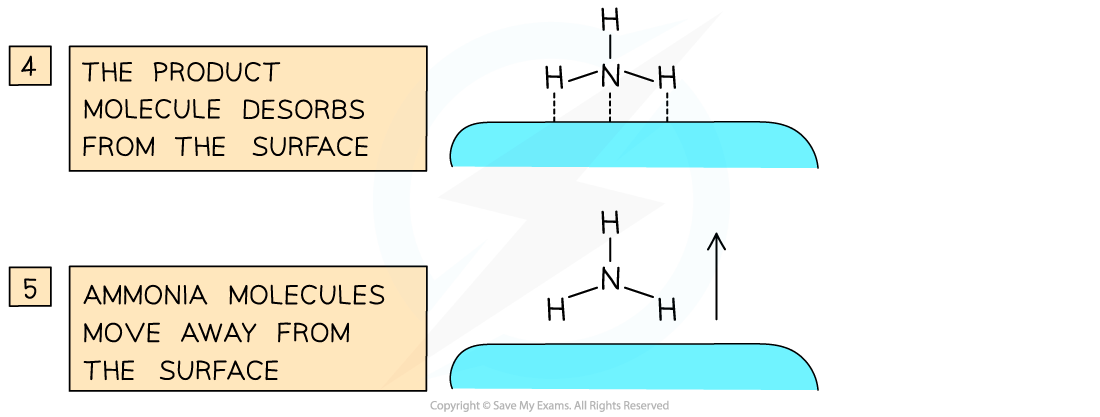
Catalysts work by attracting reactant molecules on to the surface and so providing an alternate reaction pathway of lower energy
Exam Tip
Although catalysts are not part of the overall reaction, you may see them written over the arrow in reaction equations in the same way you can add reaction conditions above or below the arrow.
轉載自savemyexams

最新發布
? 2025. All Rights Reserved. 滬ICP備2023009024號-1









