- 翰林提供學(xué)術(shù)活動(dòng)、國(guó)際課程、科研項(xiàng)目一站式留學(xué)背景提升服務(wù)!
- 400 888 0080
2015 AP Chemistry化學(xué)真題系列之簡(jiǎn)答題免費(fèi)下載
歷年AP Chemistry化學(xué)系列
真題與答案下載

翰林國(guó)際教育全網(wǎng)首發(fā)
力爭(zhēng)超快速發(fā)布最全資料
助你在升學(xué)路上一帆風(fēng)順
為你的未來(lái)保駕護(hù)航
2015 AP Chemistry Free-Response Questions Free Download
2015 AP 化學(xué)簡(jiǎn)答題部分免費(fèi)下載
考試時(shí)會(huì)提供花常用的等式與常量
以及化學(xué)元素周期表
此套Section II試卷共7題
每道大題含有不同數(shù)量的小題
共計(jì)時(shí)1小時(shí)45分鐘
其中1-3題各需23分鐘,每題10分
4-7題各需9分鐘,每題4分
完整版下載鏈接見(jiàn)文末
部分真題預(yù)覽:
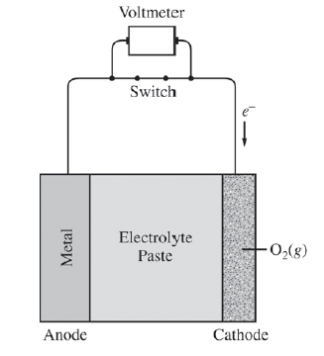
1)Metal-air cells are a relatively new type of portable energy source consisting of a metal anode, an alkaline electrolyte paste that contains water, and a porous cathode membrane that lets in oxygen from the air. A schematic of the cell is shown above. Reduction potentials for the cathode and three possible metal anodes are given in the table below.
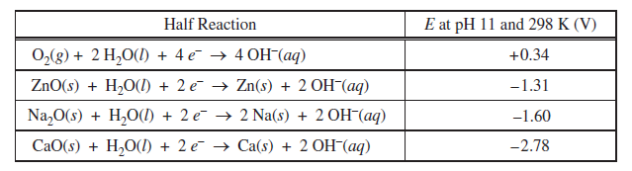
- Early forms of metal-air cells used zinc as the anode. Zinc oxide is produced as the cell operates accordingto the overall equation below.
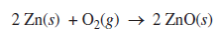
- Using the data in the table above, calculate the cell potential for the zinc-air cell.
- The electrolyte paste contains OH-?ions. On the diagram of the cell above, draw an arrow to indicate the direction of migration of OH-?ions?through the electrolyte as the cell operates.
- A fresh zinc-air cell is weighed on an analytical balance before being placed in a hearing aid for use.
- As the cell operates, does the mass of the cell increase, decrease, or remain the same?
- Justify your answer to part (b)(i) in terms of the equation for the overall cell reaction.
- ?The zinc-air cell is taken to the top of a mountain where the air pressure is lower.
- Will the cell potential be higher, lower, or the same as the cell potential at the lower elevation?
- Justify your answer to part (c)(i) based on the equation for the overall cell reaction and the information above.
- Metal-air cells need to be lightweight for many applications. In order to transfer more electrons with a
smaller mass, Na and Ca are investigated as potential anodes. A 1.0g anode of which of these metals would transfer more electrons, assuming that the anode is totally consumed during the lifetime of a cell? Justify your answer with calculations. - The only common oxide of zinc has the formula ZnO.
- ?Write the electron configuration for a Zn atom in the ground state.
- From which sublevel are electrons removed when a Zn atom in the ground state is oxidized?
完整版真題下載鏈接請(qǐng)注冊(cè)或登錄后查看
文件為PDF格式
推薦使用電腦下載
2015 AP Chemistry化學(xué)FRQ簡(jiǎn)答題完整版答案免費(fèi)下載
請(qǐng)持續(xù)關(guān)注,稍后更新
翰林學(xué)員全站資料免費(fèi)打包下載,專享高速下載通道。
[vc_btn title="查看更多AP Chemistry化學(xué)課程相關(guān)詳情" color="primary" align="center" i_icon_fontawesome="fa fa-globe" css_animation="zoomIn" button_block="true" add_icon="true" link="url:http%3A%2F%2Fwww.linstitute.net%2Farchives%2F25860||target:%20_blank|"]

最新發(fā)布
? 2025. All Rights Reserved. 滬ICP備2023009024號(hào)-1









